Project Overview
Click here to download a copy of the Lab Notebook Template for Project 1A.
Click here to download a copy of the Lab Notebook Template for Project 1B.
Click here to download a copy of the Project 1 Summary & Reflections.
Laboratory work for Project 1 will be completed in pairs. Each assignment (e.g., workshops, quizzes, notebooks) must be completed individually.
Organic chemists often try to synthesize a complex molecule from simpler starting materials. This task generally requires sequential reaction steps in which the product of one reaction serves as the reactant for the next step in the sequence. The objective of this project is to prepare the target compound (Figure 1) by a two-step reaction sequence.

Figure 1. Target compound for Project 2
The target compound is an example of a biphenyl, a compound in which two aromatic rings are bonded directly to each other. The development of synthetic methods for the production of biphenyls is important because many of these compounds exhibit important biological activity. For example, polysperlignan A (1) has demonstrated anti-neurodegenerative activity. Bicyclol (2) is an anti-hepatotoxic drug, and Merck sells the antihypertensive drug Losartan (10) (Figure 2).
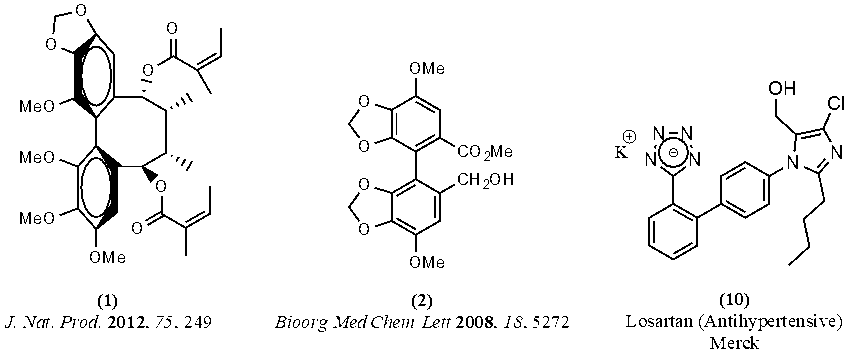
Figure 2. Some examples of biologically important biphenyls
Project 1 is intended to cultivate your skill and independence in designing and implementing a reaction sequence based on previously reported data. This is the basic work of a synthetic organic chemist. In the Project 1 scenario, you are given a “literature precedent” for each of your reactions (in the form of journal-style experimental descriptions) as well as some relevant general theoretical background. In Part A, you are expected to perform the reaction reported in the procedure at a different scale (i.e., with different quantities of materials). In Part B, you will use the literature procedure as a template for performing the same type of reaction, but you will choose substrates different than those in the reported procedure.
Remember that whenever a chemist performs a chemical reaction, there are at least three measures of success to consider:
1. What is the identity (i.e., structure) of the product(s)?
2. What is the quantity of product generated (by mass and percent yield)?
3. How pure is the isolated product?
Learning Objectives
At the end of this project, you should know how to:
• Plan and complete sequential chemical reactions based on literature precedent
• Set up and employ a reflux apparatus
• Perform a vacuum filtration
• Adjust the scale of a chemical reaction from literature precedent
• Calculate the overall yield of a reaction sequence
• Determine the substitution pattern of an aromatic ring from spectroscopic data
• Carry out an aromatic substitution reaction and a palladium-catalyzed cross-coupling reaction.
Project 1 Score Distribution
Workshop: Reaction Scaling 5%
Proficiency Quiz: Reaction Scaling 5%
Project 1A Prelab Quiz 5%
Lab Notebook 1A 30%
Workshop: Literature Templates 5%
Proficiency Quiz: Literature Templates 5%
Project 1B Prelab Quiz 5%
Lab Notebook 1B 30%
Project 1 Summary & Reflections 10%
